Salt and Polymorph Screening Services

Optimizing the Solid-State Path from Discovery to Development
At PI Health Sciences, we recognize that the selection of the correct solid form is the cornerstone of successful drug development. The physical form of your Active Pharmaceutical Ingredient (API) dictates its solubility, stability, and ultimately, its clinical performance. As a leading CRDMO, we provide integrated salt and polymorph screening services designed to de-risk your program, accelerate timelines, and build a robust intellectual property (IP) portfolio.
Our multidisciplinary team of process chemists and analytical experts works in co-located, digitally enabled labs to ensure that your molecule’s solid-state landscape is fully mapped before reaching the clinic.
The PI Health Sciences Advantage
- Integrated Solutions: Seamless transition from discovery chemistry to process R&D and commercial-scale API manufacturing.
- Scientific Leadership: Proven expertise in complex chemistries and high-potency molecules (up to OEB4).
- Sustainability (ESG): Focused on green chemistry and biocatalysis to reduce waste and energy consumption during salt/polymorph scale-up.
- Phase-Appropriate Speed: From 1g focused screens for early toxicology batches to comprehensive studies for regulatory filings.
State-of-the-Art Analytical Suite
Our solid-state characterization is supported by a global network of labs (including our sites in Hyderabad and Jaipur) equipped with 21 CFR Part 11-compliant instrumentation.
Regulatory Compliance: ICH Q6A Excellence
We ensure all solid-state data is aligned with ICH Q6A guidelines, providing the necessary justification for specifications in IND, NDA, and ANDA submissions. Our experts utilize the ICH decision tree approach to determine if polymorphic control is required based on safety, performance, and efficacy.
Our Services
Salt Screening and Selection
Enhancing Solubility and Manufacturability
Approximately 50% of marketed APIs are formulated as salts to overcome poor aqueous solubility and low bioavailability. Our tiered salt screening approach identifies the most developable ionic forms for your ionizable candidates.
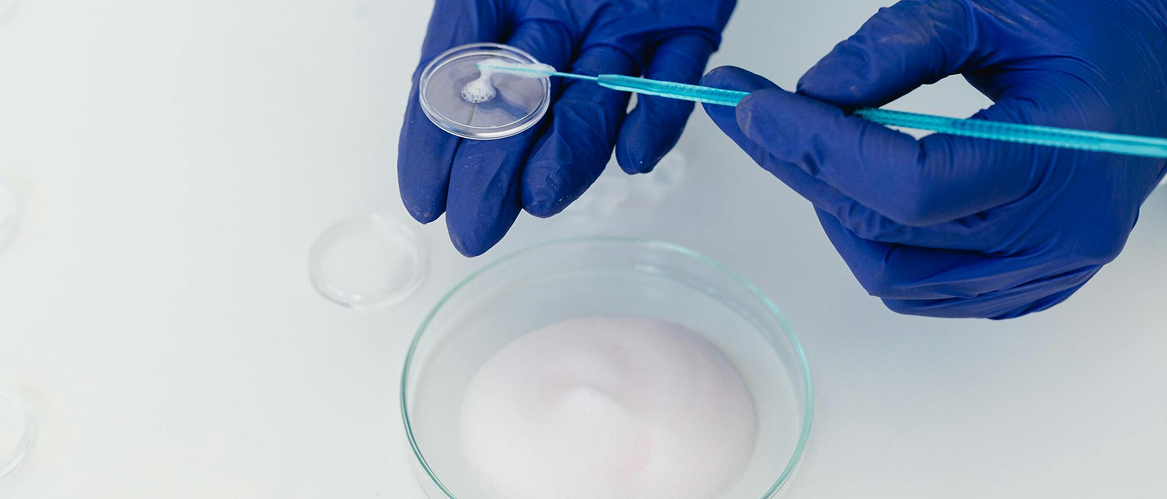
Our Approach:
- Rational Counterion Selection: We utilize a scientifically relevant library of GRAS (Generally Recognized as Safe) counterions based on $pKa$ matching (Delta pKa > 2 for stable salt formation), structural compatibility, and intended route of administration.
- High-Throughput (HT) Hit Identification: Rapid generation of salt candidates using solvent-assisted crystallization, slurry conversion, and mechanochemical grinding.
- Physicochemical Profiling: Comprehensive assessment of hits for crystallinity, non-hygroscopicity, and thermal stability to rank the most promising forms for scale-up.
Talk to Our Experts
Polymorph Screening
Enhancing Stability and IP Protection
The emergence of a "disappearing polymorph" can lead to manufacturing failures and the loss of market exclusivity. Our exhaustive polymorph screens explore the full thermodynamic and kinetic landscape of your API to ensure you are developing the most stable crystalline form.

Key Services:
- Thermodynamic Mapping: We conduct competitive slurry experiments and solvent-mediated phase transformation studies to determine the relative stability of polymorphs across various temperature ranges.
- Form Identification: Detection and characterization of anhydrous forms, hydrates, solvates, and metastable polymorphs.
- IP Expansion & Lifecycle Management: Identifying and patenting novel crystalline forms several years before patent expiration to extend product exclusivity.
- Amorphous Stabilization: For particularly challenging molecules, we provide solutions for identifying and stabilizing amorphous solid dispersions.
Talk to Our Experts
Capabilities

Thermodynamic Mapping
Determination of polymorph stability using competitive slurry experiments and solvent-mediated phase transformation studies across temperature ranges. These studies establish the relative thermodynamic hierarchy of forms, enabling identification of the most stable polymorph under process and storage conditions.


Form Identification
Detection and characterization of anhydrous forms, hydrates, solvates, and metastable polymorphs using advanced solid-state analytical techniques. Comprehensive phase identification ensures clear differentiation between closely related forms.

IP Expansion & Lifecycle Management
Identification and characterization of novel crystalline forms to support robust intellectual property strategies. By exploring and defining the solid-state landscape, we enable generation of patentable polymorphs that extend product lifecycle and strengthen market exclusivity.

Amorphous Stabilization
Development and stabilization of amorphous solid forms for poorly crystalline or low-solubility compounds. Our approach focuses on improving apparent solubility and bioavailability while addressing inherent instability risks associated with amorphous systems.
Frequently asked questions
We’re here to help with any questions you have about our plans, supported features, and how our model works.
What is salt screening in pharmaceutical development?
Salt screening is the process of evaluating different counterions that can form salts with an ionizable drug molecule. The objective is to identify a salt form that improves key physicochemical properties such as solubility, crystallinity, stability, and processability while maintaining the biological activity of the drug candidate.
Why is salt screening important during drug development?
The solid form of a drug substance can significantly influence its performance during manufacturing and formulation. Salt screening helps identify forms that provide improved stability, enhanced dissolution behavior, and better compatibility with downstream formulation strategies, reducing the risk of development challenges later in the program.
Which drug candidates typically undergo salt screening?
Salt screening is most commonly performed for small molecule drug candidates that contain ionizable functional groups capable of forming salts. Certain peptide or modified peptide molecules may also undergo salt screening when ionic interactions influence their stability or formulation behavior.
What properties are evaluated during salt screening?
Several physicochemical properties are typically examined during salt screening, including solubility, crystallinity, hygroscopicity, thermal stability, polymorphism, and dissolution characteristics. These properties help determine whether a particular salt form is suitable for pharmaceutical development.
How does salt screening influence later development stages?
Selecting an appropriate salt form early in development provides a stable foundation for formulation development, manufacturing scale-up, and regulatory documentation. A well-characterized salt form reduces the likelihood of reformulation efforts and helps ensure consistency throughout the drug development lifecycle.
Contact Us
Connect with PI Health Sciences to explore how our preclinical services de-risk development and prepare therapeutic candidates for IND submission with precision and scientific rigor.
