Integrated Discovery Biology Services

Mechanism-Driven Biology, Built for Confident Discovery Progression
PI Health Sciences Discovery Biology services offers comprehensive, end-to-end screening solutions across the full spectrum of preclinical integrated drug discovery. Backed by advanced scientific expertise and robust infrastructure, we enable seamless progression from target validation to lead selection, delivering decision-ready data to accelerate programs.
Our integrated capabilities support multiple stages of early discovery:
- Molecular target validation
- Structural and functional characterization
- Advanced biochemical and cellular screening
- ADME and pharmacokinetics profiling
- In vivo evaluation through qualified partner sites
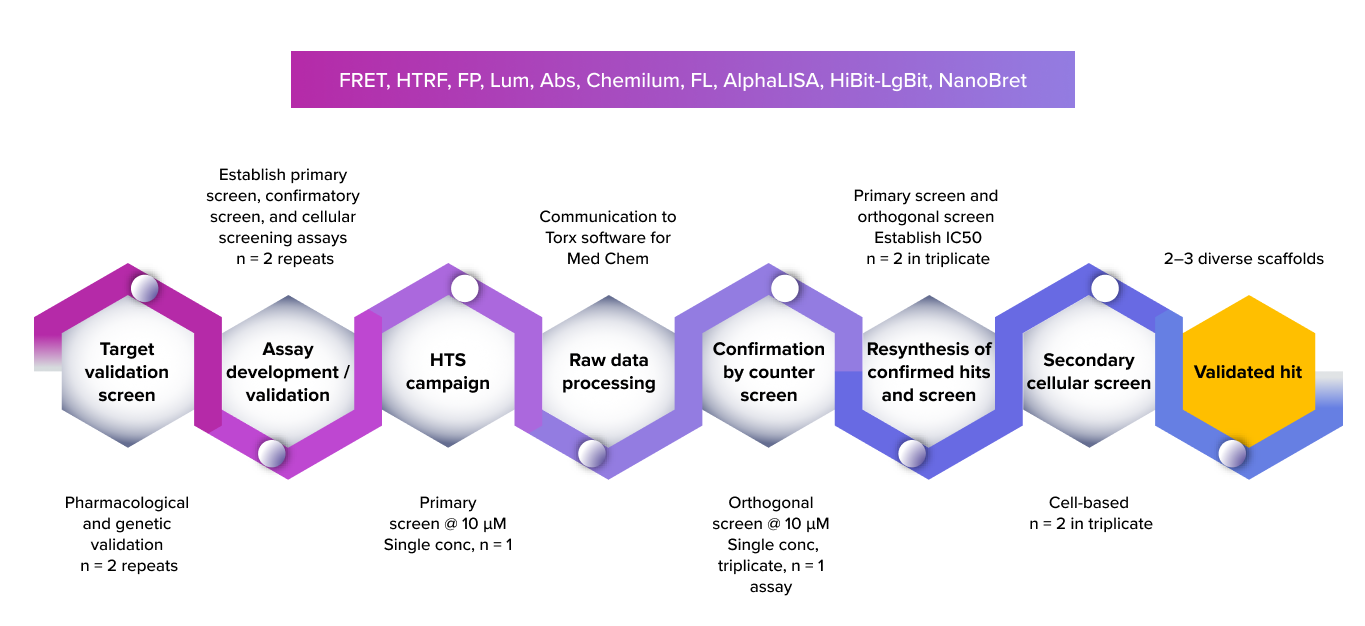
A structured HTS-to-hit validation workflow—from target and assay validation through high-throughput screening, orthogonal and cellular assays—to efficiently identify and confirm robust, diverse validated hits.
- Small molecules
- Peptides and peptide-based therapeutics
- Engineered biologics
- Emerging drug formats
- Monoclonal antibodies (mAbs)
- Antibody-drug conjugates (ADCs)
- Bi-specific antibodies
- Peptide therapeutics
Our platforms assess key biological parameters to generate high-quality outputs:
- Binding and affinity characterization
- Internalization and target engagement
- Immune-mediated functional activity
- Dose–response relationships and IC₅₀ values
Core Services
Structural Biology
Advances in genomics and high-throughput structural techniques have transformed structure- based drug discovery (SBDD), enabling faster and more precise lead identification and optimization. At PI Health Sciences, our structural biology capabilities are designed to generate high-quality protein structures that directly support rational drug design.
Our protein science team specializes in generating crystallographic-grade recombinant proteins and co-crystal structures with small-molecule ligands, enabling deep insights into binding interactions and target conformations.
We also collaborate with leading global facilities such as the Australian Synchrotron and advanced cryo-EM centers to leverage high-resolution three-dimensional structures for drug discovery and lead optimization.

Protein Production & Crystallization Services
We offer end-to-end protein generation and structural workflows to support SBDD:
- Gene cloning, plasmid preparation, and stable cell line generation
- Protein expression and purification for in vitro assays and crystallization
- Crystallization of target proteins for structure determination
Key Capabilities & Applications
Our structural biology platform enables:
- Structure-guided hit identification and lead optimization
- Co-crystal structure generation with small molecules
- Binding site characterization and interaction mapping
- Support for computational modeling and docking workflows
Why Structural Biology Matters
High-resolution structural insights help:
- Improve ligand binding affinity and selectivity
- Understand protein conformational flexibility
- Reduce design cycles through rational optimization
Talk to Our Experts
Molecular Biology
PI Health Sciences Discovery Services offers comprehensive molecular and cell engineering capabilities, supporting drug discovery programs from plasmid design to protein expression validation. Our platforms enable both prokaryotic and mammalian systems, ensuring flexibility across diverse therapeutic targets and modalities.
We specialize in generating custom engineered cell lines, with efficient timelines:
- Stable cell pool: ~4–5 weeks
- Stable cell line: ~10–12 weeks
Our expertise spans advanced transfection and delivery technologies:
- Plasmid, Lentiviral, Adenoviral, and BACMAM vector systems
- Stable and transient transfections
- Electroporation and lipid nanoparticle-based delivery
Plasmid DNA & Molecular Cloning
We provide end-to-end plasmid design and molecular cloning services, enabling reliable construct generation for downstream discovery workflows.
- Plasmid design, transformation workflows, and culture expansion
- Competent cell preparation and bacterial amplification
- Blunt-end and sticky-end cloning strategies
- Site-directed mutagenesis and advanced cloning methods
- Sequence- and ligation-free cloning approaches
Optimized systems across standard bacterial platforms:
- DH5α
- Stbl3
- JM109
Transcript Expression
Our transcript analysis workflows support target validation and pathway characterization through accurate gene expression profiling.
- RNA extraction from cell lines, tissues (fresh/frozen), and FFPE samples
- Quantitative gene expression using SYBR Green and TaqMan qPCR
- Copy number variation (CNV) analysis
- Relative expression profiling
Protein Expression & Quantification
We offer comprehensive protein expression verification and quantification using multiple orthogonal platforms for high-confidence data.
- Native PAGE and SDS-PAGE analysis
- Western blotting and automated systems (JESS)
- ELISA-based quantification
- Flow cytometry (FACS)
- Biacore-based interaction and binding analysis
- Co-immunoprecipitation for mechanistic studies
Cell Engineering
Our advanced cell engineering platforms enable precise functional and mechanistic studies in biologically relevant systems.
- Stable and transient expression systems (plasmid, viral, BACMAM)
- Electroporation and lipid nanoparticle-based delivery
- Reporter gene and promoter-based assays
- IRES-enabled multi-protein expression systems
- siRNA/shRNA-mediated gene knockdown models
Targeted and specialized expression capabilities:
- Tagged protein expression
- Organelle-specific localization (Golgi, ER, nucleus, membrane)
Talk to Our Experts
Target Identification and Validation
PI Health Sciences Discovery Biology provides integrated target identification and validation services to support early discovery programs from hypothesis generation to target confirmation. Leveraging advances in functional genomics, systems biology, and translational disease models, we enable the identification of high-confidence therapeutic targets directly linked to disease biology.
Our platforms establish target relevance, mechanism of action (MoA), and biological tractability, ensuring selected targets are:
- Biologically relevant and disease-linked
- Mechanistically validated
- Druggable and development-ready
- Aligned with downstream therapeutic strategies
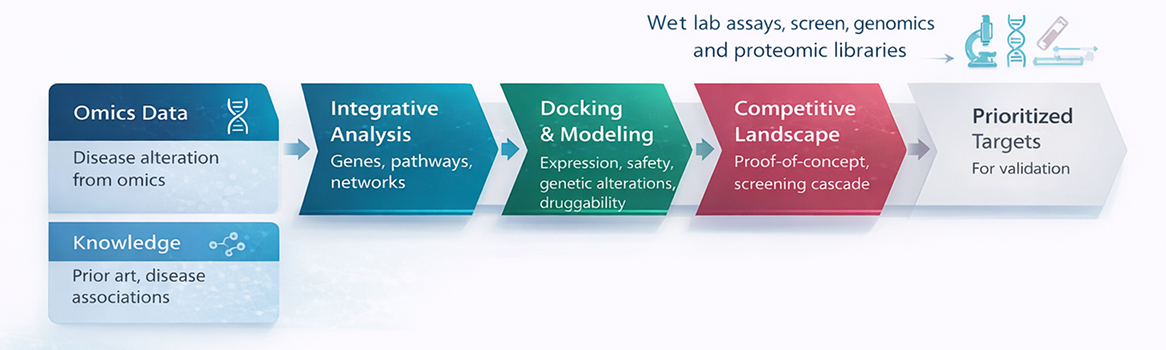
Target discovery and validation services include:
Target Selection & Characterization
We support systematic prioritization of targets through integrated biological and druggability assessment frameworks.
- Target prioritization based on disease relevance
- Druggability and tractability evaluation
- Biological characterization aligned with therapeutic strategy
- Selection of high-value targets for downstream development
Disease Mechanism Understanding
We investigate disease-driving biology to establish the causal role of targets in disease progression.
- Functional biology and pathway-linked validation
- Mechanistic studies linking targets to disease phenotype
- Evidence generation to support early discovery decisions
Model Selection & Assay Planning
We design fit-for-purpose assay systems using disease-relevant biological models.
- Cellular and molecular assay design
- Selection of appropriate disease models
- Alignment of assay readouts with validation objectives
- Scalable workflows for screening and validation
Omics (Genomics)
Our genomics-driven platforms enable systematic target identification and prioritization.
- Integrated omics data analysis
- Functional genomics-based validation
- Mapping target–disease associations
- Identification of biologically actionable targets
Pathway Analysis
We deliver mechanistic pathway validation to confirm target involvement in biological systems.
- Signaling pathway mapping and profiling
- Biomarker response and downstream effect analysis
- Functional modulation studies
- Strengthening translational relevance of targets
Knock-in/Knock-out Studies
We provide robust genetic perturbation platforms to validate target dependency and function.
- CRISPR-based knock-in and knock-out systems
- RNA interference (siRNA/shRNA) approaches
- Functional impact and pathway consequence analysis
- Mechanistic validation in cellular systems
Talk to Our Experts
In Vitro Biology Assays & Instrumentation
At PI Health Sciences, our in vitro screening and biochemical assay platforms provide the experimental backbone for integrated drug discovery across small molecules, peptides, and biologics, including monoclonal antibodies. Leveraging advanced assay technologies and scalable formats, we enable rapid hit identification, potency confirmation, and mechanistic characterization, supporting confident progression through discovery pipelines.
Our platforms are designed to deliver high-quality, decision-ready data across early discovery stages.
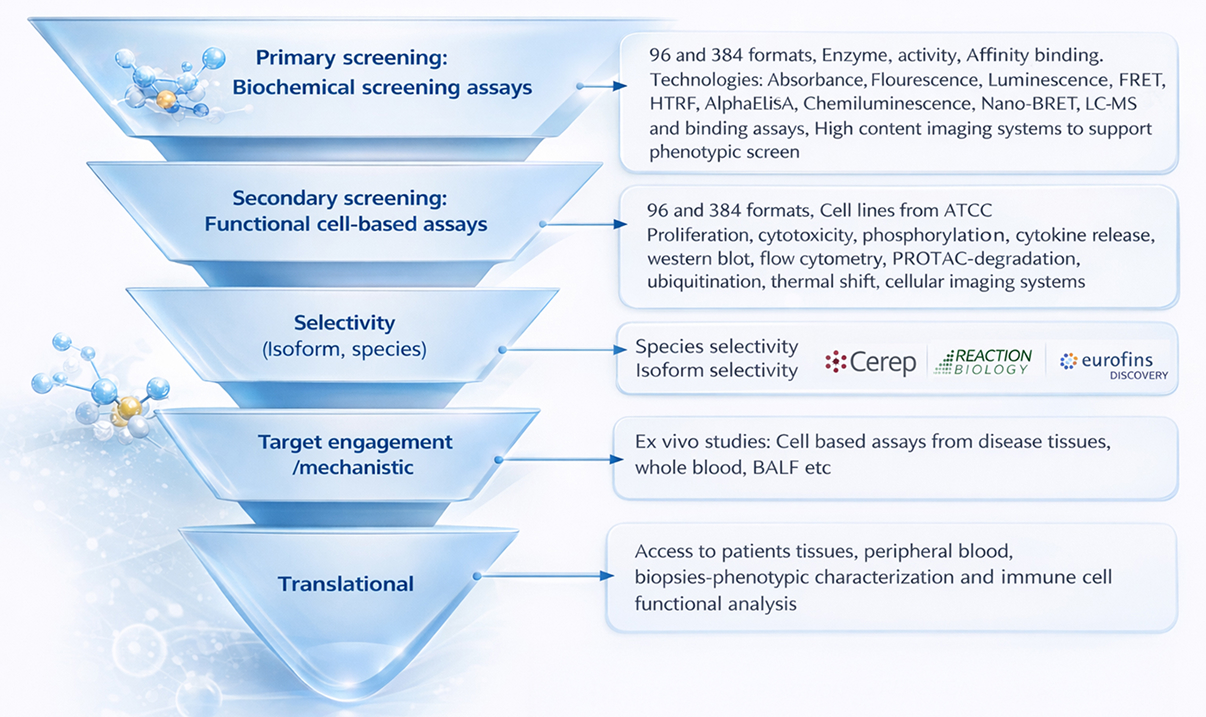
Biochemical Screening Capabilities
We offer comprehensive biochemical screening solutions to evaluate enzyme activity, binding interactions, and dose-response relationships.
- Enzyme activity and affinity-based binding assays
- Dose–response profiling and IC₅₀ determination/li>
- Percent inhibition and full curve generation
- Medium- to high-throughput screening formats
Detection technologies include:
- Fluorescence intensity and fluorescence polarization
- Time-resolved fluorescence (HTRF)
- Luminescence and absorbance
- AlphaLISA assays
- Calcium flux assays
Biophysical Interaction Analysis (SPR)
We provide quantitative Surface Plasmon Resonance (SPR)–based interaction analysis using high-throughput systems such as Biacore, enabling precise characterization of molecular interactions.
- Peptide–protein and protein–protein interactions
- Antigen–antibody binding analysis
- Small molecule–protein interaction profiling
Key outputs include:
- Association rate (k_on)
- Dissociation rate (k_off)
- Binding affinity (K_d)
- Epitope binding and relative potency assessment
Functional & Translational Screening
To enhance biological relevance, we integrate biochemical screening with cell-based and ex vivo functional assays.
- Secondary cell-based assays for cytotoxicity and proliferation
- Phosphorylation and pathway activation studies
- Cytokine release and immune functional assays
- Mechanism-of-action (MoA) validation workflows
Selectivity & Translational Profiling
We strengthen downstream confidence through selectivity and translational validation.
- Selectivity profiling across isoforms and species
- Ex vivo studies using patient-derived tissues
- Immune cell functional profiling and biomarker analysis
In Vitro Screening Services Include
- Primary biochemical screening (enzyme activity and binding assays)
- High-throughput platforms: fluorescence, luminescence, HTRF, AlphaLISA, absorbance, calcium flux
- Secondary functional cell-based assays for mechanistic validation
- SPR-based interaction analysis for kinetics, affinity, and epitope binding
- Selectivity assessment and ex vivo translational studies
Talk to Our Experts
In Vitro Pharmacology
PI Health Sciences Discovery Biology provides integrated or standalone (FFS) in vitro pharmacology services to support programs from target validation through preclinical candidate (PCC) selection. Our platforms enable comprehensive evaluation of target engagement, mechanism of action (MoA), and biomarker responses, generating decision- ready data across discovery stages.
We develop bespoke assays with validation and qualification (QC) in medium- to high- throughput formats to support compound characterization across diverse modalities.
Our capabilities include:
- Target validation and biochemical/biophysical screening
- Cell-based functional assays and MoA exploration
- Target engagement and biomarker profiling
- Assay development, validation, and QC
We bring extensive experience across targets and modalities:
- Small molecules, PROTACs, molecular glues, bicyclics
- Monoclonal and engineered antibodies
Supported therapeutic areas include:
- Oncology and Immuno-oncology
- Autoimmunity and Inflammation
- Neurologic disorders
Small Molecule Screening
We deliver scalable, high-throughput biochemical and functional screening assays to support small molecule discovery, from early hits to optimized leads.
- Enzyme activity, binding, and kinetic characterization
- Assay formats: fluorescence, absorbance, FP, luminescence
- FRET/TR-FRET, AlphaScreen, NanoBRET
- Scalable platforms: 96-, 384-, and 1536-well formats
Specialized GPCR and signaling assays:
- SPR/Biacore and FACS-based binding studies
- Functional readouts: calcium flux, cAMP, β-arrestin, IP-1
Targeted protein degradation (TPD) support:
- Binding and ternary complex assays
- Protein degradation confirmation (WB/JESS)
- Ubiquitination profiling
Large Molecule Screening
We provide integrated screening platforms for peptides, monoclonal antibodies, and engineered biologics, enabling rapid progression through early discovery.
- Affinity and kinetic profiling (Biacore 8K SPR)
- FACS-based binding assays
- ELISA-driven specificity testing
- High-content flow cytometry workflows
Therapeutic Area Specific Cell-Based Functional Assays
We design disease-relevant cellular models to support MoA exploration and translational screening across key therapeutic areas.
Autoimmunity & Inflammation
- Immune cell activation and differentiation (T, B, NK, macrophages, DCs, MDSCs, Tregs)
- Cytokine profiling and mixed lymphocyte reactions
- Cytotoxicity systems and immune functional assays
- Antibody-mediated effector assays: ADCC, CDC, ADCP
Oncology & Immuno-Oncology
- Cytotoxicity and proliferation assays (e.g., Incucyte)
- Angiogenesis, apoptosis, and invasion/motility studies
- Phosphorylation pathway analysis (WB/JESS)
- Cell cycle and cell death profiling
Tumor microenvironment (TME) models:
- Immune cell population mapping
- Checkpoint marker evaluation
- Immune–tumor co-culture systems
Fibrosis
- Epithelial–mesenchymal transition (EMT) models
- Biomarker-linked gene expression (qPCR)
Ex Vivo Immunophenotyping & Immune Profiling
To enhance translational relevance, we offer advanced ex vivo immune profiling using patient-derived and primary biological systems.
- Sample types: patient tissues, lymphoid samples, peripheral blood
- Flow cytometry-based immune subset profiling: CD4/CD8, NK, B cells, DCs, MDSCs, macrophages
- Immune activation, exhaustion, and checkpoint analysis: PD-1, TIM-3, LAG-3, OX40, 4-1BB
Functional immune readouts:
- Cytokine and chemokine profiling (ELISA, intracellular flow)
- Lymphoid tissue response assays
- T cell–dependent antibody response (TDAR) studies
Talk to Our Experts
Organoids
At PI Health Sciences, our organoid-based translational platforms enable physiologically relevant disease modeling and predictive drug response assessment beyond conventional 2D systems. By capturing tissue-level architecture, cellular heterogeneity, and functional responses, our 3D models bridge early discovery with translational biology.
These platforms support mechanism exploration, therapeutic screening, and patient-relevant validation, improving confidence in downstream decision-making.
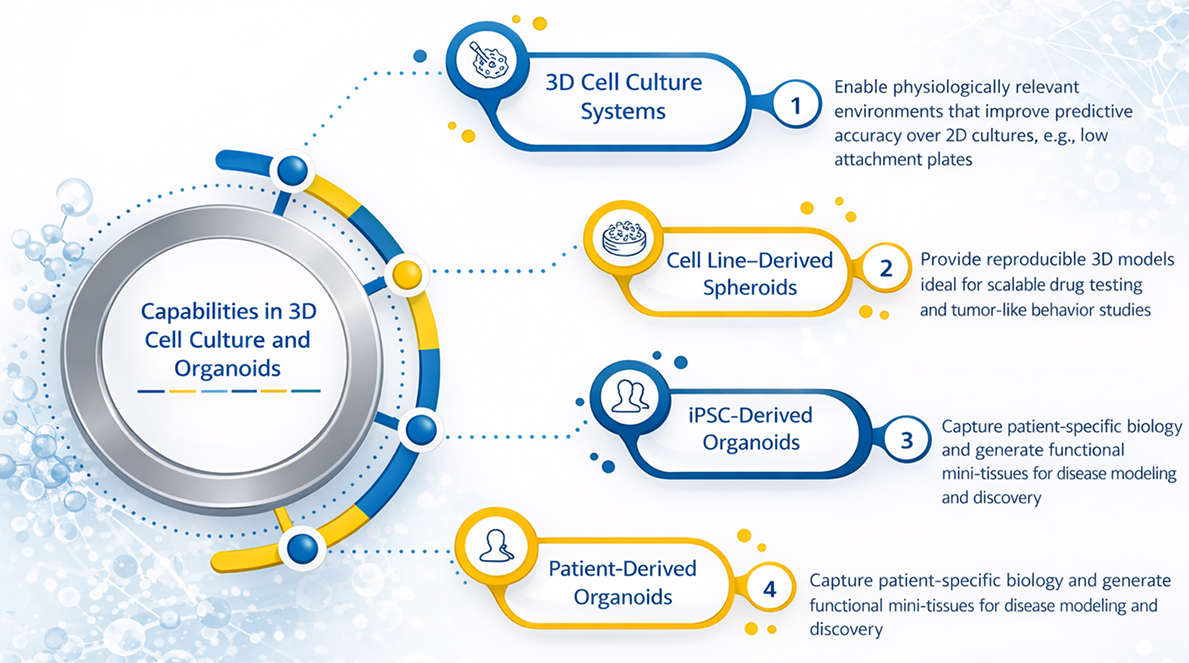
3D Modeling & Translational Capabilities
We develop scalable organoid and 3D cell culture systems to better reflect in vivo biology and enhance predictive accuracy.
- Advanced 3D cell culture systems surpassing traditional monolayer assays
- Improved physiological relevance and translational predictivity
- Support for mechanism-of-action (MoA) and functional studies
- Scalable formats for screening and validation
Organoid Platforms & Applications
Cell Line–Derived Spheroids
We generate reproducible spheroid models to enable robust and scalable drug screening.
- Tumor-like architecture and microenvironment simulation
- High-throughput drug response testing
- Reproducible systems for comparative studies
iPSC-Derived Organoids
Our induced pluripotent stem cell (iPSC)–based organoids model patient-relevant biology and disease-specific phenotypes.
- Generation of functional mini-tissues
- Disease modeling and pathway exploration
- Support for personalized and precision medicine approaches
Patient-Derived Organoids (PDOs)
We establish clinically relevant models directly from patient samples to enhance translational relevance.
- Patient-specific drug response profiling
- Personalized therapeutic evaluation
- Translational biomarker identification and validation
- Improved prediction of clinical outcomes
Talk to Our Experts
Large Molecules
At PI Health Sciences, we provide specialized Discovery Biology workflows for screening
and characterization of large molecule therapeutics, including monoclonal antibodies
(mAbs), antibody-drug conjugates (ADCs), and bi-specific platforms.
Our integrated screening cascade is designed to generate early, decision-ready evidence
across:
- Target binding and affinity strength
- Internalization and cell-surface engagement
- Immune-mediated functional activity
- Mechanistic and translational relevance
By combining scalable assay formats with biologically meaningful readouts, we enable confident selection and progression of biologic candidates across diverse therapeutic programs.
Format |
Outcomes |
Assay |
| Binding to Target Antigen | Relative Potency | Inhibition of Cell growth Assay |
| Binding Affinity and Kinetics | Binding to Target antigen by SPR | |
| Binding Activity on Cell Surface | Binding to Cell Surface Target Antigen by Flow Cytometry | |
| Phosphorylation levels | Target protein phosphorylation by Western Blot | |
| ADCC Activity | ADCC response | Primary NK cell ADCC assay |
| Reporter gene activity | FcγRIIIa Reporter Gene Assay | |
| Binding Affinity and Kinetics | Binding to FcγRIIIa 131H and 131R by SPR | |
| ADCP | Binding Affinity and Kinetics | Binding to FcγRIIa 131H and 131R by SPR |
| Apoptosis | Induction of apoptosis | Apoptosis Assay |
| Other Fcγ Receptor Binding | Binding Affinity and Kinetics | Binding to FcγRI, FcγRIIb, and FcγRIIIb by SPR |
| Additional binding assay |
FcγRIa (SPR) FcγRIa-R FcγRIa-H FcγRIIb/c FcγRIIIa-V FcγRIIIa-F FcγRIIIb |
|
| FcRn Binding | Binding Affinity and Kinetics | Binding to FcRn by SPR |
| CDC Activity | CDC Activity | CDC assay |
| Binding to C1q | C1q binding assay |
Binding Activity Assessment
PI Health Sciences initiates large molecule screening through robust binding activity assays to confirm antigen engagement and functional targeting. Our platforms support both recombinant antigen-based and cell-based binding evaluation.
Key capabilities include:
- Sandwich ELISA binding assays using Fc-tagged recombinant antigens
- FACS-based binding analysis using antigen-overexpressing CHO cell systems
- Binding assessment across varying expression levels (high, medium, low)
- Bi-specific binding assays across both tumor antigen and T-cell arms
Affinity Determination and Kinetic Profiling
To advance screening hits, we perform quantitative affinity determination using Biacore-based kinetic analysis. These studies provide critical insights into interaction strength and binding dynamics.
Services include:
- SPR-based affinity characterization of lead molecules
- Association and dissociation kinetic profiling
- Comparative ranking of candidate antibodies and constructs
Internalization and ADC Functionality Assays
For ADC and targeted biologic modalities, PI Health Sciences conducts internalization assays to quantify cellular uptake and target-driven internal trafficking, supporting payload delivery evaluation.
Capabilities include:
- High-throughput MMAF internalization assays
- Internalization kinetics using pH-sensitive dye methodologies
- Cellular uptake profiling to support ADC optimization
Immunology and Bispecific Functional Assays
For bi-specific molecules, screening extends into immune-mediated functional biology, enabling evaluation of T-cell redirection, conjugation formation, and target-specific cytotoxicity.
Key assays include:
- Bi-specific mediated T-cell and target cell conjugation analysis
- Cytotoxicity profiling in relevant co-culture systems
- Specificity assessment through selective effector cell depletion studies
Translational Extension and Cell Line Validation
PI Health Sciences further supports translational progression by extending cytotoxicity and functional assays across diverse suspension and adherent cell line models, ensuring activity consistency across clinically relevant contexts.
Applications include:
- Functional validation across multiple tumor and immune cell origins
- Translational relevance assessment beyond engineered systems
- Lead optimization support through expanded cell-based profiling
Throughput and Deliverables
Our large molecule screening platforms are scalable across:
- 24, 96, and 384-well assay formats
- Approx. ~100 molecules per week per FTE
Deliverables include:
- Percent inhibition
- Dose-response relationships
- IC₅₀ determination
- Binding, affinity, and internalization profiling
- Functional immune-mediated activity assessment
Talk to Our Experts
Therapeutic Areas
PI Health Sciences Discovery Biology delivers end-to-end assay expertise across key therapeutic areas, supporting integrated drug discovery from early screening through translational validation. Our platforms span biochemical, cellular, genomic, proteomic, and advanced tumor model systems, enabling robust evaluation across diverse therapeutic modalities.
We support:
- Small molecules, peptides
- Monoclonal antibodies, bispecifics, engineered antibodies
- Emerging therapeutic modalities
Our integrated platforms ensure mechanism-driven insights and clinically relevant data generation.
Assays
Biochemical Assay Expertise
- Kinase Activity Assays (ADP-Glo™)
- Binding and Selectivity Assays (TR-FRET, HTRF)
- Biophysical Interaction Assays (SPR, Fluorescence Polarization)
- Quantification Assays (AlphaLISA®, LANCE®)
Genomic and Proteomic Assays
- Gene Expression (qPCR, Microarray)
- Protein Expression (Western Blot, Immunofluorescence)
- Gene Silencing (siRNA/shRNA)
Analytical, In Vivo and Translational Models
- In Vivo Tumor Models (SCID Xenograft)
- Combination and Washout Studies
Ex Vivo and Translational Studies
- Immunophenotyping of TILs
- Activation Markers and Cytokine Profiling
- Transcriptome Analysis (NGS)
- Patient Tissue and Peripheral Blood Functional Immune Analysis
Immunology Assays
- Dedicated Functional Assays for:
- T Cells (Th1, Th2, Th17, CD8, NK Cells)
- Tregs
- MDSCs
- Macrophages
- Memory T Cells (CM, EM, EMRA)
- Dendritic Cells
- Immune Cell Stimulation
- Cytokine Production
- Proliferation Assays
- Antigen-Specific T Cell Recall Responses (PBMC and Animal Models)
Cellular Functional Assay Expertise
- Cell Viability and Apoptosis Assays (CellTiter-Glo®, Annexin V/PI-FACS)
- ADCC
- ADCP
- CDC
- Target Engagement Assays (HiBiT-LgBiT, NanoBRET)
Advanced Tumor Models and 3D Assays
- Amyloid-Based Hydrogels (3D Tumor Spheroid Formation)
- Biomarker Visualization (IF Microscopy)
- Live Imaging (Proliferation, Migration, Drug Response)
Immuno-Oncology In Vitro Assays
- Immune Cell Mediated Killing Assays (Small molecules, Bispecifics, Engineered Abs)
- Binding Studies (BIACORE, FACS)
- Effector:Target Co-culture Cytotoxicity Assays
- Immune Activation Marker Analysis (FACS, qPCR, NGS)
- Cytokine Release (Multiplex/Luminex)
- ADCC, ADCP
- Checkpoint Inhibitor Evaluation
- Mixed Lymphocyte Reaction (MLR)
- Biosimilar Evaluation
Talk to Our Experts
Key Strength


Therapeutic Expertise
Expertise across major disease areas including oncology, immuno-oncology, autoimmunity, inflammation, neurosciences, and fibrosis.

Laboratory & Compliance
GLP-like laboratories with plate-reader instruments compliant with 21 CFR Part 11 and computer system validation (CSV) to ensure data quality, integrity, and traceability for IND-ready submissions.


Target Classes
Comprehensive capabilities across GPCRs, kinases, phosphatases, proteases, and ion channels.
Therapeutic Modalities
Support for small molecules, PROTACs, AUTOTACs, molecular glues, and large molecules including monoclonal and engineered antibodies (in-vitro screening only).
Assay Platforms
Biochemical, biophysical, and primary cell-based phenotypic assay platforms enabling robust target and mechanism evaluation.
In-Vitro Screening Technologies
96- and 384-well screening formats covering absorbance, fluorescence, luminescence, FRET, HTRF, AlphaELISA, chemiluminescence, NanoBRET, LC-MS, and binding assays.


Imaging Capabilities
Advanced high-content imaging systems for detailed cellular and phenotypic analysis.
Frequently asked questions
We’re here to help with any questions you have about our plans, supported features, and how our model works.
What stages of drug discovery are supported by Discovery Biology services at PI Health Sciences?
Discovery Biology services supports programs from early target identification and validation through hit-to-lead and candidate selection. Our services generate biologically robust, decision-ready data to enable confident transition into preclinical development.
What therapeutic modalities are covered under Discovery Biology services?
- Small molecules
- PROTACs and molecular glues
- Peptides and bicyclics
- Monoclonal and engineered antibodies
How does Discovery Biology integrate with chemistry and DMPK programs?
Our teams operate in close alignment with medicinal chemistry and DMPK, ensuring
biological insights directly inform:
- Molecular design and optimization
- SAR interpretation
- Early liability and risk assessment
How does PI Health Sciences ensure biological data is translationally relevant?
- Disease-relevant cellular and 3D models
- Functional and mechanism-driven readouts
- Biomarker-based validation strategies
Can Discovery Biology programs be customized to specific disease areas?
Yes. Our platforms are adaptable across:
- Oncology and Immuno-oncology
- Autoimmunity and Inflammation
- Neuroscience
- Fibrosis
We design disease-specific assays with expert scientific oversight.
Contact Us
Connect with PI Health Sciences to explore how our discovery biology expertise advances target-centric programs through functional validation and biologically informed decision-making.
