As the immuno-oncology landscape continues to expand, the demand for robust, reproducible, and mechanistically informative in vitro assay systems has grown in parallel. Mixed Lymphocyte Reaction assays have emerged as one of the most relevant tools for evaluating the immunomodulatory activity of candidate therapeutics, particularly checkpoint inhibitors. At PI Health Sciences, we offer validated, high-throughput MLR assay services that provide multi-dimensional readouts- from cytokine profiling to T-cell proliferation and activation marker analysis-within a single, streamlined experimental workflow.
The Biology of Immune Checkpoints and the Rationale for MLR Assays
Immune checkpoints function as regulatory mechanisms that control T-cell activity by providing activating or inhibitory signals. These checkpoints are engaged during interactions between T-cells and antigen-presenting cells (APCs), such as dendritic cells or tumor cells. In a homeostatic context, checkpoint pathways serve to prevent autoimmunity and limit excessive immune activation. In oncological settings, however, tumor cells exploit these pathways to suppress anti-tumor immunity, rendering T-cells functionally exhausted or anergic.
Checkpoint inhibitor immunotherapies are designed to disrupt this interaction by blocking inhibitory checkpoint pathways that normally suppress T-cell function. As a result, T-cell activation is enhanced, leading to a stronger anti-tumor immune response. Several checkpoint inhibitors are currently approved for clinical use in oncology, including monoclonal antibodies targeting PD-1, PD-L1, and CTLA-4, with additional agents under active development.
The MLR assay is an in vitro system uniquely suited to interrogate checkpoint biology. In this assay, immune cells derived from two genetically distinct individuals are co-cultured to induce recognition of non-self antigens, thereby activating immune checkpoint pathways. The resulting allogeneic immune response provides an endogenous, antigen-driven T-cell stimulation context-one that closely recapitulates the T-cell, APC interactions relevant to therapeutic checkpoint modulation. This assay is commonly used to assess the immunomodulatory effects of candidate therapeutics by measuring their capacity to enhance T-cell responses.
Limitations of Conventional MLR Methodologies
Conventional methods for evaluating T-cell activation in MLR assays typically present several limitations that constrain throughput, data quality, and operational efficiency. These include:
- The need for separate assays to measure cytokine production, cell proliferation, and surface marker expression (such as ELISA and conventional flow cytometry), leading to fragmented datasets that are difficult to integrate.
- Reliance on low-throughput instruments that require relatively large sample volumes, limiting the number of test conditions evaluable per experiment.
- Manual data analysis across multiple platforms, increasing inter-analyst variability and extending turnaround time.
- Time-intensive and ardous protocols involving repeated washing steps, fixation procedures, and extensive protocol optimization.
These methodological constraints can significantly impede the pace of immunotherapy screening programs, particularly in early drug discovery where rapid iteration across multiple candidates is essential. Our MLR assay services at PI Health Sciences are specifically designed to address and overcome each of these limitations.
MLR Assay Services at PI Health Sciences: Platform Overview and Key Advantages
At PI Health Sciences, we use validated methods to perform High-Throughput Screening (HTS) MLR assays that yield a significant stimulation index (SI). Our platform has been developed and optimized to enable pharmacological evaluation of novel immunotherapies with high efficiency and data richness. The key technical advantages of our MLR platform are as follows:
1. Validated Donor Selection for Optimal Stimulation Index
A critical determinant of MLR assay performance is the degree of allogeneic mismatch between donor pairs. We use MHC class II mismatched donor pairs identified by pre-screening and validation to yield a significant stimulation index (SI) of greater than- 2.5. Donor pairs are selected based on pre-screening data to ensure robust and reproducible allogeneic responses prior to their use in pharmacological studies. This validation step ensures that the assay system is sensitive enough to detect both activating and inhibitory effects of candidate therapeutics, particularly agents with modest or graded immunomodulatory activity.
2. Multiplexed Cytokine Profiling Using Luminex Technology
Cytokine measurement is a central readout in MLR assays, providing direct evidence of T-cell activation state and the type of immune response elicited. We employ cytokine multiplexed measurements using a validated signature cytokine panel, measured using a multiplex reader (Luminex). Unlike single-plex ELISA, which can only quantify one cytokine per assay, Luminex-based multiplexing enables the simultaneous measurement of multiple cytokines, including IFN-γ, IL-2, TNF-α, IL-4, IL-6, IL-10, and others-from a single sample volume. This not only reduces sample consumption but also enables a more comprehensive characterization of the immune response profile, distinguishing Th1, Th2, regulatory, and pro-inflammatory cytokine signatures relevant to the therapeutic mechanism of interest.
3. Dye-Based T-Cell Proliferation and Activation Measurement by Flow Cytometry
T-cell proliferation is one of the most direct indicators of antigen-driven activation in the MLR system. Our platform employs dye-based measurement of cycling CD4-positive and CD8-positive T-cells through FACS-based measurement of T-cell activation and identification of recently cycling/activated CD4-positive and CD8-positive T cells using proliferation dyes (Cell Trace Dyes). CFSE (carboxyfluorescein succinimidyl ester) being a classical dye used for proliferation assays has inferior spectral separation, higher toxicity, and less even inheritance across generations. Here in PI, Cell Trace Dyes TM (CellTrace TM Violet, Cell Trace TM Red, Cell Trace TM Yellow etc.) are used for proliferation analysis. These dyes are cell-permeable fluorescent dyes that covalently bind to intracellular proteins. As cells divide, the dye is partitioned equally between daughter cells, , resulting in superior spectral separation that can be tracked by flow cytometry. This enables precise quantification of proliferation generations, allowing differentiation between actively cycling and quiescent T-cell populations.
In addition to proliferation tracking, the platform supports the measurement of activation and exhaustion markers on T-cells, as well as activation markers on APCs, providing a comprehensive picture of the immune activation state across both T-cell subsets and antigen-presenting cells.
4. Automated Data Processing for Rapid Pharmacological Evaluation
One of the core operational advantages of our MLR platform is automated data processing, allowing rapid pharmacological evaluation of novel immunotherapies and their effects on T-cell activation. Automated analysis pipelines reduce manual intervention, minimize inter-analyst variability, and shorten the time from data acquisition to pharmacological interpretation. For screening programs involving large compound libraries or multiple dose levels, automated processing is essential to maintain throughput without compromising analytical rigor.
5. PCR based evaluation of MLR
One of the unique yet useful technique is to identify the Alloreactive T-cells in the mixed culture. PCR-based methods (e.g., RT-PCR with TCRB (Clonal T cell receptor beta chain) primers) can identify specific, clonotypic T-cell responses. Besides that, PCR can detect the increase in specific genetic markers associated with rapid cell division (Ki-67, PCNA (Proliferating Cell Nuclear Antigen),TOP2A (DNA Topoisomerase II Alpha), CCND1 (Cyclin D1), CDK1 and CDK2). With PCR time kinetics for short term immune response (within hours) can also be determined.
Assay Formats: One-Way and Two-Way MLR Studies
Our MLR service offering encompasses two well-established assay formats, one-way MLR and two-way MLR-each suited to distinct experimental objectives.
One-Way MLR Study
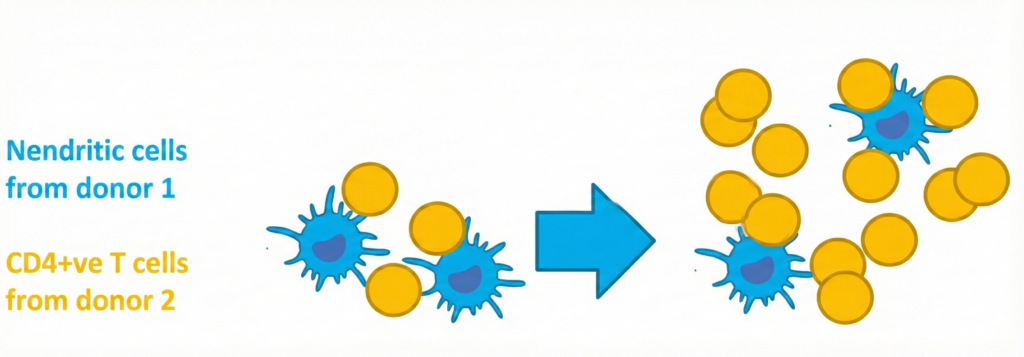
The one-way MLR study is designed to assess T-cell responses in a controlled allogeneic context where the antigen-presenting cell population is functionally inactivated. The workflow is as follows:
- Donor 1: Isolation of monocytes from peripheral blood and differentiation to dendritic cells as antigen-presenting cells (APCs).
- Donor 2: Isolation of CD4-positive T cells from peripheral blood of healthy donors.
- Co-culture in vitro in the presence of drugs and suitable controls for an optimized period of time.
Two-Way MLR Study
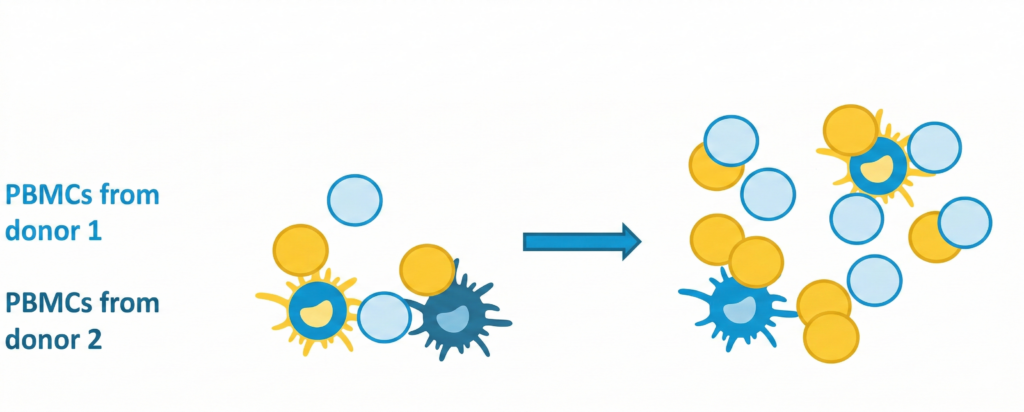
The two-way MLR study uses unfractionated PBMCs from both donors, enabling a bidirectional allogeneic immune response in which both donor cell populations serve simultaneously as responders and stimulators. The workflow involves:
- Isolation of PBMCs from fresh peripheral blood of healthy donors.
- Labelling of donor T cells using proliferation dyes (CTV, CTR etc.) to distinguish proliferating T-cell populations from each donor.
- Co-culture in the presence of drugs and suitable controls for an optimized period of time.
Scientific Applications and Therapeutic Relevance
The MLR assay platform at PI Health Sciences is optimized to support pharmacological characterization of checkpoint inhibitors, bispecific antibodies, immune cell engagers, cytokine modulators, and other immunomodulatory agents in development. By generating a significant and reproducible stimulation index (SI >3), the assay provides sufficient dynamic range to detect both enhancing and suppressive effects of therapeutic candidates on T-cell activation.
The multi-dimensional readout system- encompassing cytokine multiplexing via Luminex, FACS-based proliferation tracking, and surface marker quantification-allows for a mechanistically rich pharmacodynamic profile. This depth of characterization supports not only go/no-go decisions in early discovery but also provides mechanistic insights into the mode of action, including whether a candidate acts through cytokine-mediated pathways, direct T-cell co-stimulation, APC licensing, or checkpoint pathway blockade.
The HTS-compatible design of the platform further supports dose-response characterization across multiple concentration levels, enabling EC50 determination and comparative potency assessment across candidate panels-critical parameters in lead optimization and candidate selection workflows.
Why Partner with PI Health Sciences for MLR Assay Services?
Our MLR assay platform at PI Health Sciences represents a convergence of validated immunological methodology, high-throughput instrumentation, and rigorous data processing infrastructure. Compared to conventional MLR approaches, our system delivers:
- Integrated, multi-parameter readouts from a single assay run, eliminating the need for parallel ELISA and standalone proliferation assays.
- Validated donor pair selection based on MHC class II mismatch screening, ensuring an SI of >2 or 2.5 for reliable pharmacological discrimination.
- Luminex-based multiplexed cytokine measurement using a validated signature cytokine panel for comprehensive immune profiling.
- CTV/CTR based proliferation dye tracking with FACS readout, enabling resolution of CD4-positive and CD8-positive T-cell proliferation at single-cell resolution.
- Automated data processing pipelines for rapid pharmacological evaluation, enabling accelerated decision-making in drug discovery programs.
- Flexible assay format options, one-way and two-way MLR-to match experimental objectives and stage of program development.
Conclusion
Mixed Lymphocyte Reaction assays remain one of the most informative and physiologically grounded systems available for evaluating immunomodulatory activity in the context of T-cell checkpoint biology. Our validated, high-throughput MLR assay platform at PI Health Sciences, Genome Valley, Hyderabad, is purpose-built to meet the demands of modern immuno-oncology drug discovery-delivering reproducible, multi-dimensional pharmacodynamic data that supports candidate selection, mechanism of action characterization, and lead optimization.
To learn more about our MLR assay capabilities or to discuss how our platform can support your immunotherapy program, contact our Discovery Biology and Translational Sciences team at PI Health Sciences.

