In Drug discovery and development, the precise quantification of biomolecular interactions, including binding kinetics, affinity constants, and concentration measurements-constitutes a foundational requirement for candidate characterization and lead optimization. Surface Plasmon Resonance (SPR) assays has established itself as the gold-standard technique for real-time, label-free measurement of these interactions, offering unparalleled mechanistic resolution at the molecular level.
At PI Health Sciences, our Discovery Biology services and Translational Sciences division offers comprehensive SPR assay services, encompassing peptide-protein, protein-protein, antigen-antibody, and protein-small molecule interaction studies. Powered by the high-throughput Biacore 8K platform, our SPR services deliver quantitative kinetic and affinity data with rapid turnaround times, supporting programs from early hit identification through to late-stage candidate characterization.
The Physical Principles of Surface Plasmon Resonance
Surface Plasmon Resonance is an optical sensing technique based on the excitation of collective electron oscillations at the interface between a conductive metal and a dielectric medium upon illumination with polarized light. Under resonance conditions, incident photons couple to surface plasmon polaritons, producing an evanescent electromagnetic field that propagates along the metal-dielectric boundary. The resonance condition is highly dependent on the refractive index of the dielectric environment adjacent to the metal surface.
In SPR measurements, polarized light is directed toward a thin metal film, typically gold, through a coupling medium that satisfies momentum-matching requirements. At a defined incident angle or wavelength, energy transfer from the incident light to surface plasmons results in a characteristic reduction in reflected light intensity. Molecular binding events at the sensor surface modify the local refractive index, leading to measurable shifts in the resonance angle or wavelength.
Analysis of these shifts enables real-time, label-free quantification of biomolecular interactions, including association and dissociation kinetics, equilibrium binding constants, and analyte concentrations. The evanescent field penetrates only a few hundred nanometers beyond the sensor surface, ensuring that signal changes are exclusively attributable to binding events at the surface rather than bulk solution effects. This physical property confers high specificity and sensitivity to SPR measurements, making it highly suited for the quantitative study of molecular interactions relevant to drug discovery.
Instrument Platform: Biacore 8K, High-Throughput SPR for Parallel Assay Development
Our SPR assay services are delivered on the Biacore 8K platform, a purpose-engineered high-throughput SPR system. The Biacore 8K is equipped with eight needles and 16 flow-cells, providing a high-throughput platform for parallel assay development with a sample capacity of up to 4 × 384-well microplates. This configuration enables simultaneous analysis of multiple analyte-ligand combinations within a single experimental run, dramatically increasing throughput without sacrificing data quality.
The instrument is supported with GxP complaint machine-learning enabled software. This enhances the accuracy and ensures complete data integrity. The Insight software also allows parallel processing of samples and data analysis modules thereby, further cutting down the turnover around time of the studies.
SPR Assay Portfolio: Interaction Types, Reagent Requirements, and Turnaround Times
Our SPR service offering encompasses four core interaction categories, each with defined reagent requirements and expected turnaround times:
| Assay Type | Reagent Requirements | Output Data | Turnaround Time |
| Peptide-protein interaction | 10–20 µg | Kon, Koff, KD | 21-25 days |
| Protein-protein interaction detection | ~100 µg – 200 µg (depending upon nature of protein) | Kon, Koff, KD | 28 days |
| Antigen-antibody interaction detection | Antibody: ~50–100 µg; Antigen: ~100–200 µg | Kon, Koff, KD | 28 days |
| Protein-small molecule interaction detection | Protein: ~100–200 µg; Small molecule: ~100–200 µg | Kon, Koff, KD | 28 days |
Assay Deliverables: Quantitative Kinetic and Affinity Parameters
For each SPR study conducted at PI Health Sciences, the following deliverables are generated:
- Association rate constant (kon): The rate at which the analyte binds to the immobilized ligand on the sensor surface, expressed in units of M⁻¹s⁻¹. A higher kon indicates faster binding onset.
- Dissociation rate constant (koff): The rate at which the analyte dissociates from the ligand, expressed in units of s⁻¹. A lower koff indicates a more stable complex with slower dissociation.
- Equilibrium dissociation constant (KD): Calculated as the ratio koff/kon, the KD provides a thermodynamic measure of binding affinity. A lower KD indicates higher binding affinity.
- Generation of information on titer, concentration, and kinetics of target in a single assay cycle: The platform is designed to extract maximal informational content per experimental run, enabling the simultaneous determination of binding concentration, kinetic parameters, and activity titer within a single acquisition cycle.
These parameters are fundamental to multiple stages of the drug discovery process: KD values inform potency benchmarking and structure-activity relationship (SAR) analysis; kon and koff individually contribute to understanding the kinetic selectivity profile of candidates; and concentration measurements support quality control of reagents and analytes used across programs.
Analytical Modes: Screening, Kinetics, Affinity, Epitope Binning, Concentration, and Relative Potency
The Biacore 8K platform at PI Health Sciences supports a wide repertoire of analytical modes tailored to distinct drug discovery objectives:
Screening
In a screening mode, large panels of candidate molecules are tested against an immobilized target to identify binders. The high-throughput capacity of the Biacore 8K – accommodating up to 4 × 384-well microplates – makes it well-suited to primary screen formats where rapid identification of active hits is the priority.
Kinetics
Kinetic analysis involves fitting the association and dissociation phases of SPR sensorgrams to appropriate binding models (e.g., 1:1 Langmuir model) to extract kon and koff with high precision. This mode is particularly valuable for lead optimization, where subtle changes in molecular structure can translate into meaningful differences in binding kinetics.
Affinity
Affinity determination via equilibrium analysis provides KD values even for interactions with very fast kinetics where rate constant separation may be difficult. Steady-state affinity measurements complement kinetic data and are often used in parallel to cross-validate results.
Epitope Binning
Epitope binning experiments determine whether two antibodies or ligands compete for the same binding site on a target molecule. This analysis is particularly relevant for antibody drug discovery and biologics characterization, where understanding epitope coverage across a panel of candidates informs selection of molecules with non-redundant mechanisms of action.
Concentration
Active concentration determination by SPR measures the fraction of analyte in a sample that is functionally capable of binding its target, providing a more biologically meaningful measure of sample activity than total protein concentration determined by absorbance or colorimetric methods.
Relative Potency
Relative potency assessment by SPR enables side-by-side comparison of candidate molecules against a reference standard, providing quantitative potency ratios that support batch release, comparability studies, and candidate rank-ordering in lead optimization campaigns.
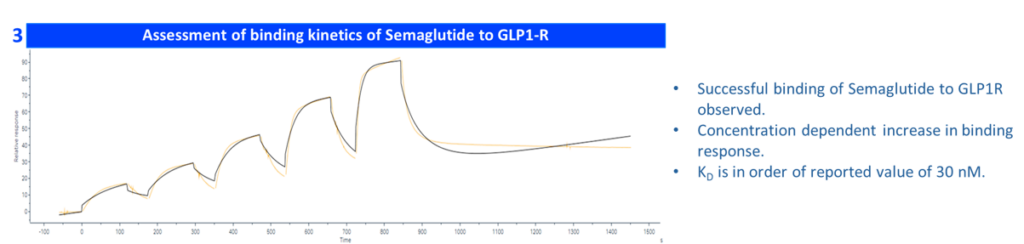
Case Study: Binding Kinetics of Semaglutide with GLP-1 Receptor Using SPR Methodology (Biacore 8K)
This case study also illustrates the informational depth achievable from a single SPR experiment: titer, concentration, and kinetic parameters are generated within a single assay cycle, providing a comprehensive interaction profile with minimal sample consumption and rapid turnaround.
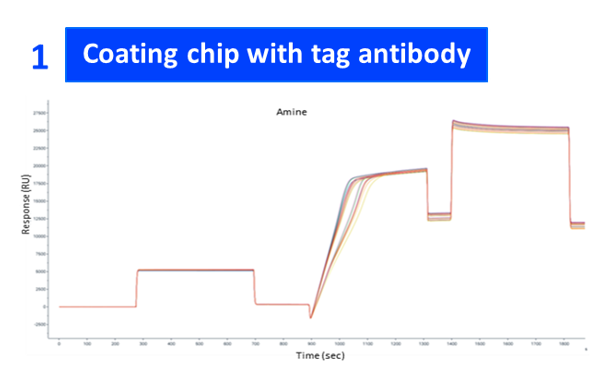
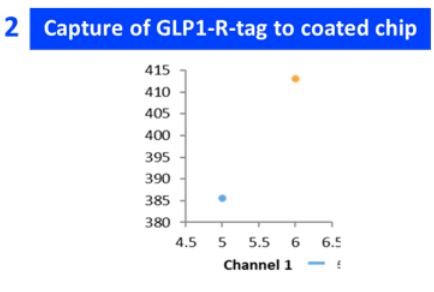
| GLP1R conc | Semaglutide conc (nM) | General kinetics model | 1:1 binding kₐ (1/M·s) | Kd (1/s) | Kᴅ (M) |
| 4 µg/ml | 6.25, 12.5, 25, 50, 100 | 1:1 binding | 6.14E+08 | 2.49E+00 | 3.30E-08 |
Why Partner with PI Health Sciences for SPR Assay Services?
Our SPR assay services at PI Health Sciences are designed to meet the technical demands of modern drug discovery programs across a broad spectrum of molecular interaction types. Key differentiators of our SPR platform include:
- High-throughput capability: Eight needles and 16 flow-cells on the Biacore 8K enable parallel assay development with sample capacity for up to 4 × 384-well microplates, supporting large screening campaigns.
- Comprehensive interaction coverage: Peptide-protein, protein-protein, antigen-antibody, and protein-small molecule interaction studies are all within scope.
- Multi-parameter outputs: Each study delivers kon, koff, and KD, along with titer, concentration, and kinetic data-all from a single assay cycle.
- Multiple analytical modes: Screening, kinetics, affinity, epitope binning, concentration, and relative potency-all supported by a single platform.
- Defined turnaround times: From 21-25 days for peptide-protein studies to 28 days for protein-protein, antigen-antibody, and small molecule interaction studies-providing program predictability.
Conclusion
SPR Assay remains an indispensable analytical technology in drug discovery, providing unmatched real-time, label-free resolution of biomolecular interaction kinetics and affinity across diverse molecular classes. Our SPR assay services at PI Health Sciences, delivered on the Biacore 8K platform, offer a scientifically rigorous, high-throughput, and quantitatively comprehensive solution for the characterization of molecular interactions across peptide-protein, protein-protein, antigen-antibody, and protein-small molecule systems.
Whether you require primary screening of compound libraries, detailed kinetic characterization of lead candidates, epitope binning of antibody panels, or active concentration determination for biologics, our SPR platform is equipped to deliver the data. Contact PI health sciences for advance program development for your needs with confidence.

